AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Meaning of resonance in chemistry8/13/2023 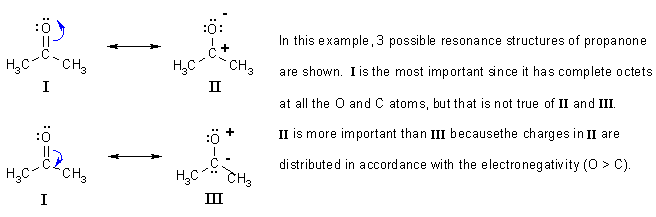
Resonance hybrids must have the same number of unpaired electrons (if exists). Resonance hybrids cannot be made to have lower energy than the actual molecules.ĥ. (iii) The resonating structures must have the same number of paired and unpaired electrons. When separating charge (giving rise to ions), usually structures have little contribution where negative charges are on less electronegative elements, but this may not be true if additional bonds are gained.Ĥ. (ii) Resonance occurs only when all the atoms lie in the same plane. The Total number of electrons and total charge must be constant.ģ. Resonance theory explain various observed properties in a molecule using electron delocalization and multiple Lewis. Position of nuclei must be the same in all structures otherwise they will form isomers.Ģ. If the actual energy of the molecule is E 0 which is determined experimentally then:ġ. Understanding resonance in chemistry helps in figuring out the stability and energy states of a compound. A complex Lewis structure is referred to as resonance. Also, it occurs through the interaction of two pi bonds in adjacent atoms. Each individual Lewis structure is called a contributing structure of the target molecule or ion. From these three energies, E 3 is having lowest energy which corresponds to the most stable resonating structure. The resonance effect refers to how an electron lone pair interacts with a pi bond to form polarity in a molecule. Resonance Definition: Resonance is a method of describing the delocalized electrons in some molecules where the bonding cannot be explicitly expressed by a single Lewis structure. The symbols M or R are used to represent the resonance effect. Let us suppose that a molecule have three resonating structures which have energy E 1, E 2, E 3 associated with them. The withdrawal or releasing effect of electrons attributed to a specific substituent through the delocalization of or pi-electrons, which can be seen by drawing different canonical structures, is known as a resonance or mesomeric effect. The difference between the actual energy of the molecule and that of most stable resonating structure is known as Resonance Energy.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
